 
The theory was given by Sidgwick and Powell in the year 1940. Polar molecules usually have an asymmetrical shape and their net dipole moment is never zero. Electronegativity difference is the main factor due to which polarity occurs in a molecule. Consequently, the bond dipole moments do not cancel one another, and the result is a molecule which has a dipole moment. As such, VSEPR is an extremely powerful tool, because molecular shape offers insight into a wide range of important physical properties (polarity, solubility. The VSEPR theory is a tool that is used for predicting the shape of a molecule from the electron pairs that surround the central atoms of that molecule. If we look up this notation in the VSEPR chart given below we find that PO43- has a tetrahedral shape. However, although a molecule like CHCl 3 has a tetrahedral geometry, the atoms bonded to carbon are not identical. In contrast, \(CHCl_3\) is a polar molecule (right panel in figure above). 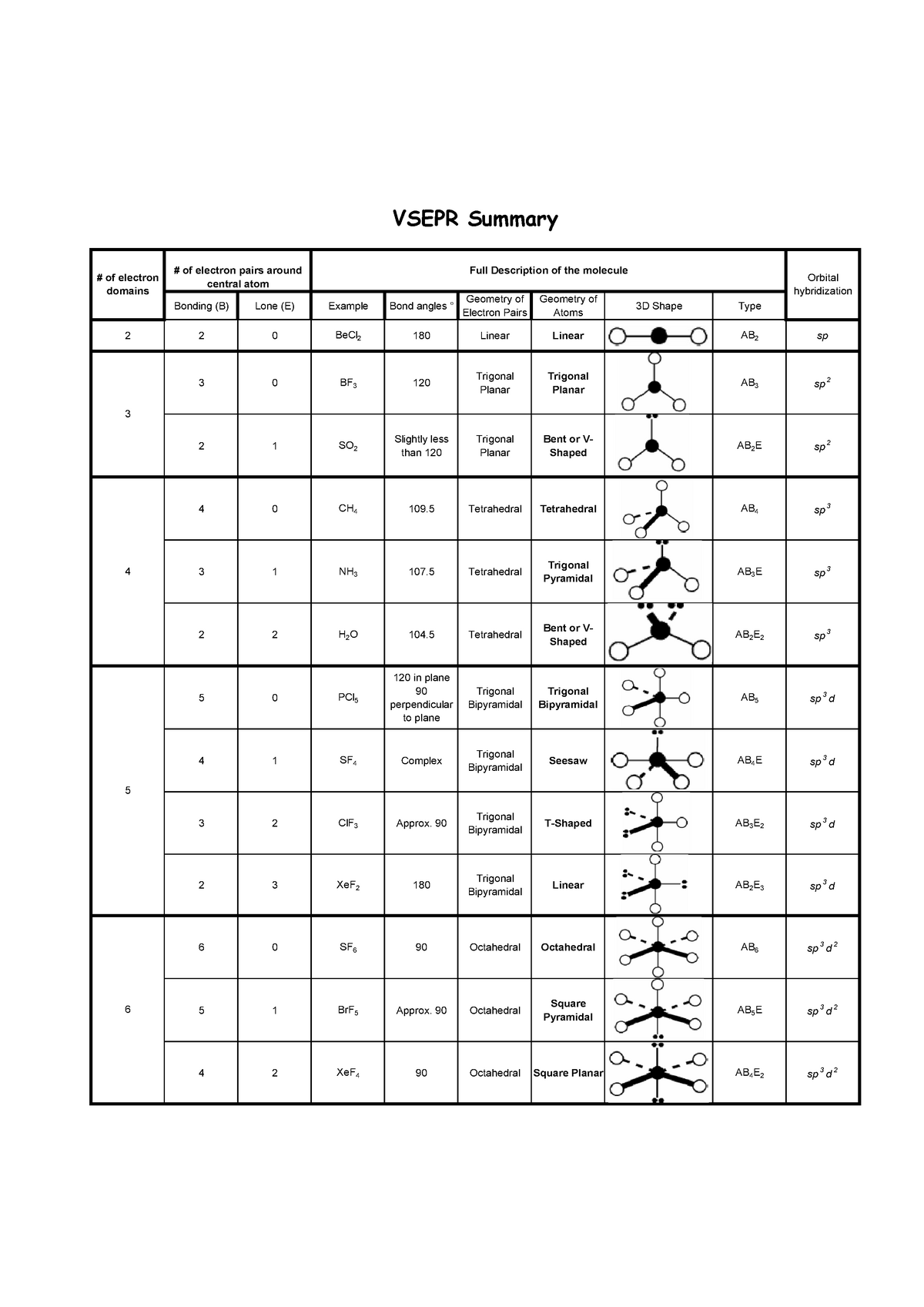
The whole of the outside of the molecule is somewhat negative, but there is no overall separation of charge from top to bottom, or from left to right. The VSEPR structures take the names of 3-D geometric shapes, as in the example trigonal bipyramidal. 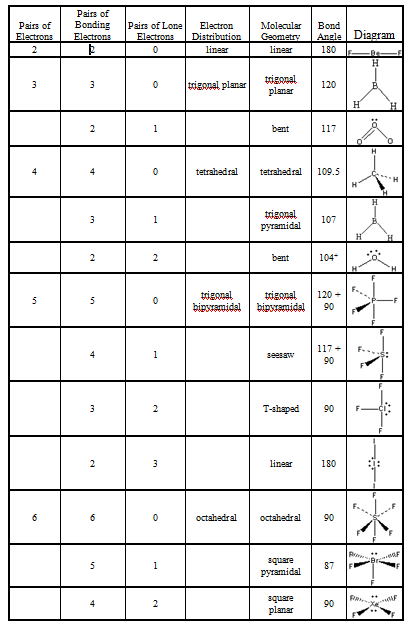
The electronegative chlorine draws electrons towards itself.Ĭonsider \(CCl_4\), (left panel in figure below), which as a molecule is not polar - in the sense that it doesn't have an end (or a side) which is slightly negative and one which is slightly positive. The manner in which the individual bonds contribute to the dipole moment of the molecule is nicely illustrated by the series of chloromethanes shown below. In more complex molecules with more than one polar covalent bonds, the three-dimensional geometry and the compound’s symmetry determine whether the molecule has a net dipole moment. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
